This policy brief was originally published in Japanese as part of a series of papers produced by a joint research project conducted by the Japan Center for International Exchange (JCIE) and the Tokyo University Institute for Future Initiatives (IFI) to provide analyses on global and regional health governance systems and structures and to offer concrete recommendations about the role Japan should play in the field of global health.
The need for an international framework for sharing pathogens in response to pandemics
Since the onset of the global COVID-19 pandemic, there has been a vigorous debate aimed at restructuring global health governance. One issue that must be considered in that context is the mechanism for rapid international sharing of pathogens and related information, namely the genetic sequence data (GSD). The need for such a mechanism has been raised on numerous occasions, including by the World Health Organization (WHO) Independent Panel for Pandemic Preparedness and Response (IPPPR),1 at the Global Preparedness Monitoring Board (GPMB),2 in the declaration by G7 health ministers,3 at the G20 High Level Independent Panel,4 and at the World Health Assembly in November 2021.5
The existing framework allowing international sharing of influenza viruses with human pandemic potential (IVPP) dates back to the outbreak of the avian influenza bird flu A (H5N1) in 2007, when Indonesia decided to withhold sharing samples of the virus, citing its sovereignty over genetic resources within its territory, as recognized by the Convention on Biodiversity (CBD). This refusal to share the pathogen prompted discussions at the WHO that led to the establishment of the Pandemic Influenza Preparedness (PIP) Framework. This framework, however, was limited to IVPP and did not cover other pathogens. For that reason, ever since then, the need for an international sharing framework for other pathogens has been raised each time another international outbreak of infectious disease has occurred.
This paper addresses the issues and options of international frameworks on sharing of pathogens and GSD based on the review of existing literature and other sources.6 Although pathogens and GSD are closely related, they each nevertheless have different qualities; therefore, they are discussed separately in this paper (see table 1). For pathogens, for instance, special facilities and procedures are required, in addition, international bodies have already developed frameworks and rules regarding access and benefit-sharing (ABS) for genetic resources to a certain extent. On the other hand, no similar frameworks exist yet for GSD— and these are precisely the issues that are currently being discussed. It is also necessary to consider the fact that GSD could potentially serve as an alternative for pathogens in the future.
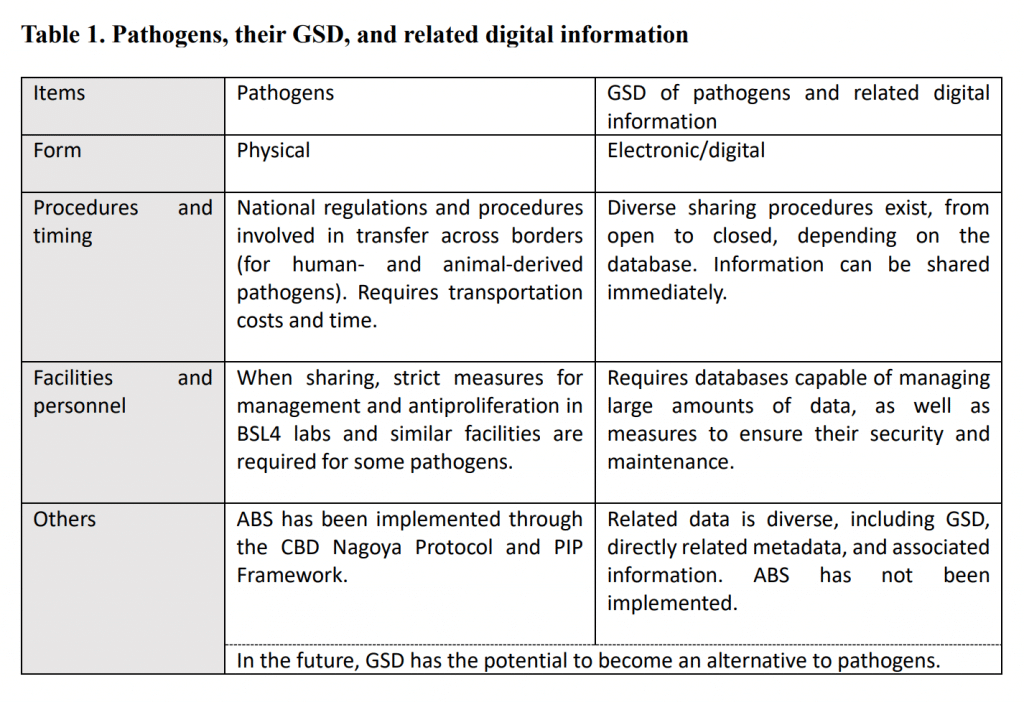
International frameworks and issues related to pathogen sharing
Sharing pathogens involves transfer across national borders. International frameworks related to this, apart from those whose purpose is public health, include the Convention on Biological Diversity (CBD) and its supplementary agreement, the Nagoya Protocol on Access to Genetic Resources and the Fair and Equitable Sharing of Benefits Arising from their Utilization (ABS). As is well known, in addition to the preservation and sustainable use of biological diversity, the objectives of the CBD include fair and balanced ABS with regard to the benefits arising from the use of genetic resources. Viruses and other pathogens are included in the scope of the CBD’s definition of “genetic resources,” and are therefore covered by the ABS mechanism (meaning that any use requires prior consent and mutual agreement). Procedures for ABS under the CBD are stipulated in the Nagoya Protocol. This contains provisions stating that the protocol does not apply in cases where other specialized international frameworks already exist and are consistent with the objectives of the protocol (Article 4.4), and provisions regarding special considerations for public health (Article 8 (b)).
In responding to a pandemic, it may be possible to use Article 4.4, for example, to exempt pathogens covered by the PIP Framework and other frameworks from being covered by the Nagoya Protocol. However, each country has its own interpretation of whether the PIP Framework qualifies under this provision when incorporating the protocol into their domestic law. In Japan, PIP Framework and strains of seasonal influenza for producing vaccines are excluded from the ABS guidelines of the Nagoya Protocol, but this is not necessarily the case in other countries. As a result, complex regulations that differ from country to country are impeding sharing procedures.
From the perspective of public health, the WHO’s International Health Regulations (IHR) and the PIP Framework mentioned above could also be relevant. The IHR requires member states to provide public health information to the WHO in the case of a public health emergency of international concern (PHEIC). At present, however, pathogens and GSD are not included in this requirement. On the other hand, the PIP Framework makes international sharing possible regarding IVPP, and ABS frameworks also exist for commercial uses of IVPP.7 However, as mentioned above, this applies only to IVPP, and does not include the novel coronaviruses or other pathogens. Also, unlike the IHR, this framework is not legally binding.
In addition to the frameworks and entities mentioned above from the perspective of biological resources and public health, there is a wide range of organizations and systems in other fields that can also be considered in the context of pathogen sharing, including the UN Food and Agriculture Organization (FAO) and the World Organization of Animal Health (OIE) from the perspective of zoonotic diseases and the One Health approach; the World Trade Organization (WTO) from the perspective of intellectual property in vaccine research and development; and the Biological Weapons Convention (BWC) from the perspective of biosecurity. Coordination with the international bodies that have jurisdiction over these organizations is another potential topic for consideration.
Next, let us review some of the issues that have been raised to date with the international sharing of pathogens.8 In addition to the uncertainty regarding measures and procedures taken by individual countries with regard to ABS in the CBD and Nagoya Protocol for reasons of sovereignty over viruses, another political factor that has been raised involves cases where a state may be reluctant to acknowledge the presence of pathogens in order to protect its reputation and out of fear of the potential impact on trade and travel. In low-and-middle-income countries, a lack of capacity in terms of human resources, facilities, and financial resources may make sharing impossible regardless of intent. Furthermore, from a biosecurity perspective, strict procedures are needed with pathogens that could cause a pandemic. Ethical considerations and the protection of personal data are also important factors. The norm within the scientific community has been for the sharing of pathogens to be carried out on a trust basis; for this reason, many feel that the procedures required by the Nagoya Protocol and other regulations impose a heavy burden. On the other hand, criticisms have also been leveled at the scientific community. Since information on pathogens is not disclosed while peer review is still underway, some people have claimed that the peer review process for scientific journals is obstructing the prompt sharing of information.9 Another issue is that the “benefits” at stake in ABS are decided by mutual agreement between the parties involved. Since these benefits exist in different forms, both monetary and non-monetary, reaching such an agreement can take time. Also, the sharing of pathogens is currently done on an ad hoc, bilateral basis; there is no framework for efficient sharing on a multilateral basis.
Potential international frameworks for GSD and issues involved in its sharing
With regard to GDS, discussions are ongoing in international bodies on definitions, scope, and the handling of such data. In the context of the Nagoya Protocol, the term “digital sequence information” (DSI) is being used in place of GSD in discussions. In developing countries in particular, it is argued that DSI should also be covered by the ABS guidelines. But since the CBD defines genetic resources as “materials,” and since a vast database of GSD already exists that has been openly shared, differences of opinion on this matter have become evident between developing countries on the one side and developed countries and industry on the other.10 Meanwhile, issues involving GSD are also being discussed at the WHO. Noteworthy recent progress on this front includes the decision at the 70th World Health Assembly (WHA) in 2017 to consider approaches to GSD within the PIP Framework and the subsequent publication of reports based on that decision.
In addition to the issues listed above affecting pathogen sharing and similar issues, one problem that can be cited in relation to the sharing of GSD in particular is the lack of experience with ABS. Although there is an understanding that the benefits arising from the use of GSD should be shared appropriately and fairly, various issues would arise if the ABS mechanism was extended to include GSD. In particular, the scientific community has concerns about the use of open data and the possible impact on research. There is also a need to consider how to track usage of digital data, which differs from pathogens that can be managed physically. Another complicating factor is the fact that various data analysis organizations have been constructed historically, each with their own regulations on data disclosure; differences in usage limitations, conditions, and targets; different rules regarding permission to reuse and process data; and so on. Another point that will need to be considered is the fact that, as mentioned at the outset, GSD could be used as an alternative to pathogens in the future. At present, pathogens are still essential for uses such as making test kits and developing vaccines, and GSD cannot act as a substitute in those cases, but as technology develops, concerns about ABS and biosecurity will become more pressing, and it would be better to prepare for that eventuality by considering our response now.
An examination of international sharing of pathogens
Finally, policy options for the international sharing of pathogens are considered below (fig. 1), taking into consideration the points summarized above. Increasingly, GSD and pathogens are discussed all together in the international debate, however, since each has its own issues, as described above, I will focus here on the sharing of pathogens.
There are generally two possible directions that can be taken: either focus on improving existing international frameworks or build new ones. In the case of the former approach, there would be a need to take into consideration all the debates to date within the WHO and CBD. If the framework of the WHO is chosen, one possibility (Option 1) would be to revise the IHR to include pathogens in the public health information that member states are obliged to report to the WHO (IHR Article 6.2). Since the IHR are legally binding on all member states, this would have an advantage in terms of effectiveness, although some people are skeptical, noting that since the IHR are not fully functional even now, it is questionable whether expanding the scope of reporting requirements would have the desired effect. If a slightly more moderate framework were to be chosen, one conceivable option would be to use apply the PIP Framework. Specifically, this would involve expanding the framework to cover not only influenza viruses but also other pathogens capable of causing a pandemic (Option 2). The advantages of this option would be that the Global Influenza Surveillance and Response System (GISRS) already exists as a network for collecting and sharing information on IVPP and seasonal influenza, and that the structure for and experience with ABS exists for IVPP. In addition, some sharing of data on COVID-19 has already begun using the PIP Framework platform.11 On the other hand, because the PIP Framework is not legally binding, countries do not necessarily share pathogens, and this might diminish the effectiveness of this approach. There was discussion in the past about expanding the framework to include pathogens other than IVPP, and at that time there were concerns that doing so might risk exceeding the capacity of the GISRS. Therefore, if this option were to be pursued, it would be necessary to consider questions of funding and operational capacity at the same time. In transferring pathogens across national borders, it is likely that the CBD Nagoya Protocol will be involved regardless of which option is chosen, and there would also be a need for coordination with the provisions of the protocol (as addressed below, in our discussion of Option 4).
Next, there is the possibility of using the CBD framework (Option 3). But considering a lack of global health expertise and the fact that the United States, one of the major countries, is not a signatory, it would not be realistic to address the issue using the CBD framework alone. As mentioned above, however, since the CBD Nagoya Protocol contains provisions that take into consideration other international frameworks and public health concerns (Article 4.4 and Article 8 (b)), one possible approach would be to consider using the CBD in collaboration with the framework of the WHO or other organizations (Option 4). In concrete terms, this approach would involve producing guidance and guidelines for the interpretation of the related articles of the Nagoya Protocol mentioned above in order to promote harmonization of domestic measures on the handling of pathogens that currently differ among countries (or have not yet been stipulated). In parallel, steps could be taken to expand the framework of the WHO’s PIP Framework to include pathogens, or a new framework could be drawn up to incorporate them (Option 5). As already noted, Japan has excluded the PIP Framework and seasonal influenza strains for vaccine production from the ABS guidelines of the Nagoya Protocol, but in fact few signatory states have taken such clear measures using domestic legislation. It may be possible to use the Japanese domestic law as a model to work toward harmonization.
One option that might be envisaged as a new framework (Option 5) is the pandemic treaty or other instruments currently being discussed, with European countries playing a leading role. This framework could offer the possibility of incorporating Option 4. Those who support this approach argue that it would be quicker to create a new framework than to devote energy to improving existing dysfunctional frameworks. This may be true to a certain degree, but even the new framework approach was selected, it would be necessary to think about how to coordinate with and enhance existing frameworks, and the advantages would need to outweigh the merits of using existing frameworks. Both approaches would require long negotiations among countries to turn them into a reality. One approach that aims to make gradual progress is the WHO BioHub System that was established in May 2021 by the WHO and the Swiss government. This is now operating on a trial basis, limited to non-commercial sharing among member states who can reach an agreement.12 If this type of system is to be expanded to function fully in the future, it nonetheless will have to coordinate with existing frameworks at some stage.
This policy brief has summarized international frameworks related to pathogens and GSD and issues related to their international sharing based on the existing literature, and has carried out some preliminary discussions of these issues. Such an analysis requires that we also consider a variety of factors, including the individual interests of the countries involved, time constraints, feasibility, and political momentum. In addition, although GSD was dealt with separately in this brief, the positioning of GSD within international frameworks is itself an issue that needs to be considered in the future. While following the developments in various international discussions, we must further examine how to achieve global sharing of pathogens and GSD in order to contribute to improved public health in the international community.
About the Author
Makiko Matsuo is Project Associate Professor of the Graduate School of Public Policy at the University of Tokyo.
Notes
- Independent Panel for Pandemic Preparedness and Response, COVID-19: Make it the Last Pandemic, https://theindependentpanel.org/wp-content/uploads/2021/05/COVID-19-Make-it-the-Last-Pandemic_final.pdf.
- Global Preparedness Monitoring Board, A World Prepared—Global Preparedness Monitoring Board Strategic Plan 2021-2023 (2021), https://www.gpmb.org/docs/librariesprovider17/default-document-library/gpmbstrategicplan-2021-23.pdf.
- G7 Carbis Bay Health Declaration (2021), Ministry of Foreign Affairs of Japan website, https://www.mofa.go.jp/files/100200011.pdf.
- G20 High Level Independent Panel on Financing the Global Commons for Pandemic Preparedness and Response, A Global Deal for Our Pandemic Age (2021), https://pandemic-financing.org/report/foreword/.
- The topic was raised in a paper discussed at a special session at the WHO held in November 2021, 2021, in the context of developing a “WHO Convention, Agreement or Other International Instrument. See WHO, “Draft Report of the Member States Working Group on Strengthening WHO Preparedness and Response to Health Emergencies to the special session of the World Health Assembly (2021), https://apps.who.int/gb/wgpr/pdf_files/wgpr5/A_WGPR5_2-en.pdf.
- This section draws largely on Burci et al., “International Sharing of Human Pathogens to Promote Global Health Security—Still a Work in Progress,” ASIL Insights 25, no. 13 (2021); Rizk et al., “Everybody Knows This Needs to Be Done, But Nobody Really Wants to Do It,” Graduate Institute of International and Development Studies, Global Health Centre (2020); Rourke et al., “Policy Opportunities to Enhance Sharing for Pandemic Research,” Science 368, no. 6492(2020): 716–18; Gostin et al., “The Global Health Law Trilogy: Towards a Safer, Healthier, and Fairer World,” Lancet 390, no. 10105 (2017): 1918–26; Centre for EvidenceBased Medicine, “WHO Consultation on Data and Results Sharing During Public Health Emergencies— Background Briefing” (2015).
- Where a private company is involved in the PIPF, the company signs an SMTA2 of the Standard Material Transfer Agreement (SMTA), agreeing to partnership contribution and to provide benefits to the WHO. The CBD Nagoya Protocol may be subject to domestic legislation in some countries.
- See note 6.
- It has been pointed out, however, that during the ongoing COVID-19 pandemic, there has been an explosive increase in the publication of pre-prints that have not yet undergone peer review.
- For details on the Nagoya Protocol, see Junji Ichihara, Naoto Koyama, and Keiko Nozaki, “Seibutsu tayosei joyaku posuto-2020 seibusu tayosei wakugumi Dai-3-kai kokai sagyo bukai (online), sanka hokoku Dijitaru hairetsu joho o chushin ni,” (Report from the Third Meeting of the Working Group on the Convention on Biological Diversity: Post-2020 Frameworks on Biological Diversity (Online), Focusing on Digital Sequence Information), in Bioscience and Industry 79, no. 6 (2021): 498–501.
- WHO, “Preventing the Next Human Influenza Pandemic: Celebrating 10 years of the Pandemic Influenza 8 Preparedness Framework,” https://www.who.int/news/item/21-05-2021-preventing-the-next-human-influenzapandemic-celebrating-10-years-of-the-pandemic-influenza-preparedness-framework.
- Efforts have started on a trial basis to share information on the coronavirus for non-commercial purposes among member states who can reach an agreement on cooperation. See the WHO website, https://www.who.int/initiatives/who-biohub.

